All published articles of this journal are available on ScienceDirect.
Efficacy of Linezolid as a Novel Therapeutic Agent against Toxoplasma gondii RH Strain: In Vitro and BALB/c Mouse Models
Abstract
Introduction/Background
Toxoplasma gondii (T. gondii) is an important human and veterinary pathogen, and current therapies such as sulfadiazine and pyrimethamine may cause adverse effects. Identifying alternative treatments is therefore essential. This study aimed to evaluate the antiparasitic activity of the antibiotic linezolid against Toxoplasma gondii in vitro and in vivo.
Materials and Methods
Linezolid and standard drugs were prepared at concentrations ranging from 0.625 to 40 µg/mL and tested against tachyzoite using microscopic examination, cell viability assays, and flow cytometry. Cytotoxicity was assessed in HeLa cells. For the in vivo experiment, BALB/c mice were infected with T. gondii and treated with linezolid for 14 days. Survival was monitored and compared to untreated controls and to standard drug treatment.
Results
Linezolid reduced tachyzoite counts in vitro in a time-dependent manner (p<0.05). The calculated IC50 was 12.4 µg/mL, while cytotoxicity on HeLa cells (CC50) was 92.3 µg/mL, indicating moderate selectivity (SI=7.4). Flow cytometry showed induction of early and late apoptosis in tachyzoites. In vivo, untreated mice died by day nine, while linezolid therapy prolonged survival to day eleven. Combination treatment with pyrimethamine and linezolid resulted in the longest survival, reaching day 13.
Discussion
These findings suggest that linezolid inhibits parasite growth and triggers apoptotic death with limited toxicity to host cells. Although the in vivo survival benefit was modest and comparable to standard therapy, combination treatment enhanced overall efficacy.
Conclusion
Linezolid shows potential as an adjunct or alternative therapy for toxoplasmosis and warrants further investigation in optimized dosing regimens and combination approaches.
1. INTRODUCTION
Toxoplasma gondii (T. gondii) is an obligate intracellular protozoan parasite that infects a wide range of warm-blooded animals, including humans [1]. It is estimated that Toxoplasma gondii infection affects approximately one-third of the human population [2]. T. gondii has a complex life cycle that includes a definitive host (cats) and an intermediate host (including humans). Infection can occur by ingestion of oocysts from contaminated food or water, congenital transmission, or organ transplantation [3, 4]. The clinical manifestations of toxoplasmosis vary widely, ranging from asymptomatic cases to severe disease characterized by encephalitis, chorioretinitis, and systemic complications [5]. T. gondii can cause severe health complications, especially in immunocompromised individuals, pregnant women, and newborns [6]. Current treatments for T. gondii infection primarily include pyrimethamine and sulfadiazine, often combined with leucovorin to reduce hematologic toxicity. However, these treatments have limitations, including side effects, contraindications in pregnant women, and the emergence of drug-resistant strains [7] and resistance problems. Reports have indicated that novel therapeutic strategies are needed to combat the increasing burden of toxoplasmosis, especially in immunocompromised populations [8]. Recent interest has focused on drug repurposing, especially antibiotics that target parasite organelles resembling prokaryotic systems.
Most of the available studies have focused on traditional antiparasitic therapies, leaving the potential of linezolid largely unknown. Linezolid is an oxazolidinone antibiotic primarily used for the treatment of multidrug-resistant Gram-positive bacterial infections. Its mechanism of action involves inhibition of bacterial protein synthesis [9]. This antibiotic inhibits bacterial protein synthesis by binding to the 50S ribosomal subunit. T. gondii contains two organelles with prokaryotic-like ribosomes: the apicoplast and the mitochondrion. This structural similarity may explain the observed antiparasitic activity, as inhibition of mitochondrial or apicoplast translation can disrupt parasite replication and survival. Recent studies have shown that linezolid may have antiparasitic activity, particularly against certain protozoa, which has increased interest in its potential repurposing for the treatment of T. gondii. Preliminary in vitro studies have shown that linezolid has activity against T. gondii, suggesting its potential as a therapeutic candidate [10]. The rationale for investigating linezolid against T. gondii is twofold: first, its unique mechanism of action could provide a novel approach to overcome limitations associated with existing antiparasitic therapies. Second, repurposing an already approved drug could accelerate the availability of effective treatments for T. gondii infections. Furthermore, evaluating the efficacy of linezolid in a well-established BALB/c mouse model allows for the evaluation of both the in vivo and in vitro effects of the drug on T. gondii and helps to understand its therapeutic potential and safety profile.
The aim of this study was to evaluate the efficacy and potential therapeutic effects of linezolid in the treatment of infections caused by T. gondii. This included in vivo experiments using BALB/c mice and in vitro studies to assess the efficacy, safety, and mechanism of action of the drug against the parasite.
2. MATERIALS AND METHODS
2.1. Ethics Statement
The study was done according to the institutional animal ethics guidelines, which were approved by the Ethical Committee of the Tarbiat Modares University with ethics number IR.MODARES.AEC.1402.065. The source of the animals is Royan Laboratory Animal Science Center, Tehran, Iran.
2.2. Host Cells and Parasite Strain
HeLa cells were cultured in DMEM (Dulbecco's Modified Eagle Medium) or RPMI (Roswell Park Memorial Institute) medium containing 10% FBS (Fetal Bovine Serum) and antibiotics (penicillin and streptomycin). The cells were passaged using trypsin/EDTA (Ethylenediaminetetraacetic acid), and the culture medium was changed every 24 hours to maintain optimal growth conditions [11].
T. gondii tachyzoites were obtained from the Parasitology Department of the University of Tehran. Tachyzoites were diluted in phosphate-buffered saline (PBS) and maintained by intraperitoneal passages (IP) in female BALB/c mice. To infect mice, a solution containing an initial concentration of 2×106T. gondii tachyzoites was prepared. 0.5 cc of this solution was injected intraperitoneally into each BALB/c mouse. This method was used to maintain the parasite strain and prevent attenuation, as tachyzoites were continuously passed through the peritoneum of BALB/c mice for different passages.
2.3. Drug Preparations
Linezolid (Samen –Iran), sulfadiazine (Sigma-Aldrich, USA), and pyrimethamine (Sigma-Aldrich, USA) were purchased. Sulfadiazine and pyrimethamine were prepared at concentrations of 40 μg/mL and 2 μg/mL. Linezolid was used in a concentration range from 0.625 μg/mL to 40 μg/mL for in vitro experiments. All prepared solutions were stored at -20°C.
2.4. Viability Assessment of T. gondii Tachyzoites
For the assessment of the viability of T. gondii tachyzoites, a total of 2×10^6 tachyzoites were treated with varying concentrations of linezolid (0.625, 1.25, 2.5, 5, 10, 20, and 40 μg/ml) over three different time points: 3, 6, and 24 hours post-treatment.Each experiment was in triplicate and in three independent replications. To differentiate between live and dead parasites, a combination of staining techniques and microscopic evaluation was employed. Specifically, the trypan blue exclusion assay, which allows for the identification of viable tachyzoites was utilized. Live parasites remain unstained by trypan blue, while dead parasites take up the dye and appear blue under microscopy. The parasite count was done by using a hemocytometer.
For quantification, the number of live and dead tachyzoites using a Neuberger counting chamber (Neobar slide) was counted. The percentage of viability was also calculated by the formula (1):
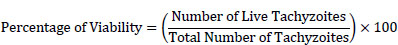
This approach made it possible to accurately evaluate the effect of linezolid on the viability of T. gondii tachyzoites during the treatment period.
2.5. Treatment of HeLa Cells Infected with T. gondii
HeLa cells were infected with T. gondii using a parasite-to-cell ratio of 5:1. After allowing parasites to attach for 3 hours, linezolid was administered at concentrations of 2.5, 5, 10, 20, and 40 μg/mL. Cells were incubated for 24 hours, fixed with methanol, and stained with Giemsa stain for subsequent analysis.
2.6. Cytotoxicity Assay
For the toxicity survey, 100 μL of RPMI medium supplemented with 10% FBS was added to each well of a 96-well plate. Each well was then treated with Linezolid at concentrations ranging from 0.625 μg/mL to 40 μg/mL, as well as sulfadiazine and pyrimethamine at concentrations of 40 μg/mL and 2 μg/mL. Following this, 100 μL of cultured HeLa cells, approximately 1 x 105 cells/mL, were added to all wells. To ensure accurate assessment, three wells were included as negative controls, which contained only RPMI medium with 10% FBS and no drug treatment. All plates were incubated at 37 °C for 24 hours. Subsequently, MTT (3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyl tetrazolium bromide; Merck Chemical Company, Germany) was employed to evaluate the viability of the HeLa cells. The MTT reagent was dissolved in PBS at a concentration of 5 mg/mL and filtered to remove any particulates. Then, 10 μL of the prepared MTT solution was added to each well, and the plates were incubated at 37 °C for 4 hours. Following incubation, DMSO was added to all wells to dissolve the formazan crystals, and the absorbance was measured using an ELISA reader at a wavelength of 570 nm [12].
2.7. Flow Cytometry
T. gondii tachyzoites (2×106) were distributed into three separate microtubes and treated with linezolid (concentrations of 5 and 10 μg/ml), for 24 h at 37°C. Controls did not receive any drug. After the incubation period, the samples were prepared and transferred to a research center (Azma Baft Company) for flow cytometry (Annexin V/PI staining), and the results were analyzed using Cell Quest software [13].
2.8. In vivo Study
In this study, female BALB/c miceaged 6-8 weeks and weighing 20-25g were categorized into four groups (n = 5 mice in each group).Animals were housed in standard aluminum cages in the laboratory at a temperature of 20-23°C and a 12-hour light-dark cycle. All groups were injected subcutaneously with 1×103T. gondii tachyzoites to induce infection. The experimental groups included: The mice received no treatment (control group), sulfadiazine +pyrimethamine (group II), linezolid (group III), and pyrimethamine+linezolid (group IV). The treatment consisted of 5 injections of the drugs, one day apart, as follows: control group (the mice received no treatment), sulfadiazine +pyrimethamine 100 μl of pyrimethamine (2 mg/kg/day) and sulfadiazine (40 mg/kg/day), linezolid (100 μl of 25 mg/kg/day), and pyrimethamine+linezolid (100 μl of 0.2µg/ml) injected intraperitonially for 14 days. The survival periods of all mice were monitored daily. The significant differences in survival rate were evaluated by the Kaplan–Meier test.
2.9. Statistical Analysis
To find out the normality of the data distribution, the Kolmogorov-Smirnov test was used. Descriptive statistics were also used to summarize the baseline characteristics of the participants. The comparison of the laboratory parameters between the intervention and control groups was performed using a One-Way ANOVA test to compare means across groups. The Tukey post hoc test was employed for pairwise comparison of differences. Moreover, SPSS Version 26 software was used for data analysis, and GraphPad PRISM 2.0 software was also utilized for the graphical representation of results. The statistical analyses were performed at a two-sided ≤0.05 significance level.
3. RESULTS
3.1. In vitro Results
3.1.1. Inhibition of Tachyzoite Growth
After 3, 6, and 24 hours of parasite culture, parasite counts showed that the number of tachyzoites decreased in the presence of different concentrations of linezolid (Fig. 1). In addition, a statistically significant difference was observed between the different concentrations of the drug and the control group at these three different times studied (p<0.5). The results of this difference are shown in Tables 1, 2, and 3, respectively. The calculated IC50 was 12.4 µg/mL, while cytotoxicity on HeLa cells (CC50) was 92.3 µg/mL, indicating moderate selectivity (SI=7.4).
3.1.2. Flow Cytometry Analysis
Flow cytometry results at different concentrations of linezolid, 5 μg/mL and 10 μg/mL, as well as the control group, have been depicted in Figs. (2, 3, and 4), respectively. In these figures, the percentages of tachyzoites undergoing primary apoptosis (bottom right), secondary apoptosis (top right), necrosis (top left), and viable cells (bottom left) have been shown.
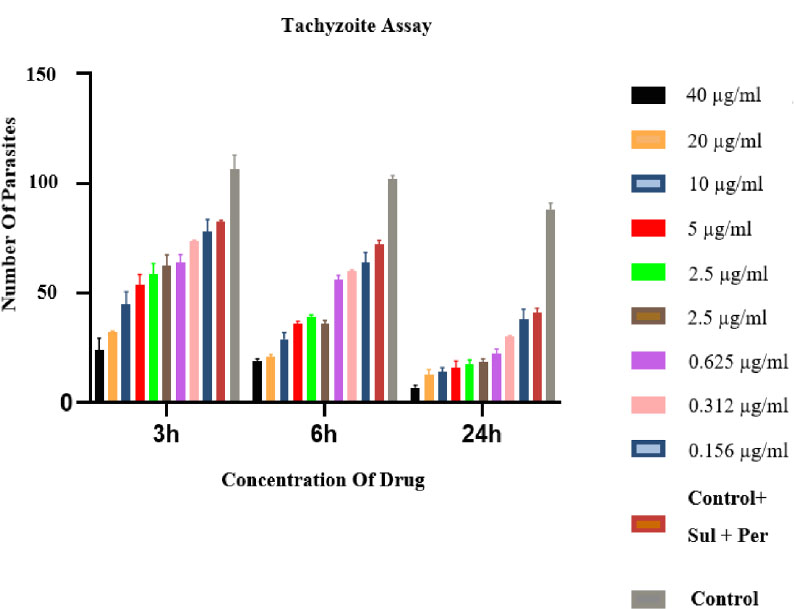
Effect of different concentrations of linezolid on the number of tachyzoite after the various cultivation times. There are statistical differences for all treated groups in comparison to the negative control group p<0.05.
| Group No. |
Group Name [Linezolid Con. (μg/mL)] |
Viable Tachyzoite No. (Mean ± SD*) |
Groups with Statistically Significant Differences (≤0.05) |
|---|---|---|---|
| 1 | 40 | 24±5.6 | 11-4 |
| 2 | 20 | 32±0.7 | 11-4 |
| 3 | 10 | 45±5.6 | 1, 8-11 |
| 4 | 5 | 53.5 ±4.9 | 1-2, 8-11 |
| 5 | 2.5 | 58.5±4.9 | 1-2, 8-11 |
| 6 | 1.25 | 62.5±4.94 | 1-2, 10-11 |
| 7 | 0.625 | 64±3.53 | 1-2, 10-11 |
| 8 | 0.312 | 73.5±0.7 | 1-4, 11 |
| 9 | 0.156 | 78±5.6 | 1-4, 11 |
| 10 | Control+ (Sul + Per) | 82.5±0.7 | 1-7, 11 |
| 11 | Control- | 106.5±6.36 | 1-10 |
| Group No. |
Group Name [Linezolid Con. (μg/mL)] |
Viable Tachyzoite No. (Mean ± SD*) |
Groups with Statistically Significant Differences (≤0.05) |
|---|---|---|---|
| 1 | 40 | 19±1 | 11-4 |
| 2 | 20 | 21±1 | 11-4 |
| 3 | 10 | 29±3 | 1, 8-11 |
| 4 | 5 | 36±1 | 1-2, 8-11 |
| 5 | 2.5 | 39±1 | 1-2, 8-11 |
| 6 | 1.25 | 36±1.5 | 1-2, 10-11 |
| 7 | 0.625 | 56±2 | 1-2, 10-11 |
| 8 | 0.312 | 60±0.5 | 1-4, 11 |
| 9 | 0.156 | 64±4.5 | 1-4, 11 |
| 10 | Control+ (Sul + Per) | 72±2 | 1-7, 11 |
| 11 | Control- | 102±1.5 | 1-10 |
| Group No. |
Group Name [Linezolid Con. (μg/mL)] |
Viable Tachyzoite No. (Mean ± SD*) |
Groups with Statistically Significant Differences (≤0.05) |
|---|---|---|---|
| 1 | 40 | 7 ±1 | 11-4 |
| 2 | 20 | 12.5±2.5 | 11-4 |
| 3 | 10 | 14±2 | 1, 8-11 |
| 4 | 5 | 16±3 | 1-2, 8-11 |
| 5 | 2.5 | 17.5±2 | 1-2, 8-11 |
| 6 | 1.25 | 18.5±1.5 | 1-2, 10-11 |
| 7 | 0.625 | 22.5±2 | 1-2, 10-11 |
| 8 | 0.312 | 30±0.5 | 1-4, 11 |
| 9 | 0.156 | 38±4.5 | 1-4, 11 |
| 10 | Control+ (Sul + Per) | 41±2 | 1-7, 11 |
| 11 | Control- | 88 ±3 | 1-10 |
3.1.3. MTT Assay
The survival percentage at the different concentrations of linezolid in uninfected HeLa cells 24 hours after culture is shown in Fig. (5). No statistically significant difference was observed between the treated and control groups (p=0.099). Since the p-value is greater than 0.05, the difference between the drug-treated and control groups is not statistically significant.
3.1.4. Infected Cells and Number of Tachyzoite in HeLa Cells
The results from cell culture showed that at higher concentrations of linezolid, the number of tachyzoites decreased (Fig. 6). In addition, HeLa cells were viable in the culture media, which could be seen under the microscope (Fig. 6). The percentage of parasite-infected cells 24 h after drug exposure compared to the control groups is demonstrated in Fig. (7). Additionally, the mean and standard deviation of tachyzoite counts in treated and control cells after 24 h are illustrated in Fig. (8).
3.2. In vivo Results
Monitoring and examination were conducted across all groups until the end of day 14. The survival rate in the negative control group indicated that all mice died by the ninth day postinfection. The most favorable outcome was observed in the group treated with pyrimethamine and sulfadiazine, where one mouse survived until the tenth day. Following this, the linezolid group had one mouse that lived until the eleventh day. Lastly, in the group receiving both pyrimethamine and linezolid, one mouse survived up to the thirteenth day. According to the Kaplan–Meier test, the survival rate was statistically different from the control group. The survival rates across the various groups are illustrated in Fig. (9).
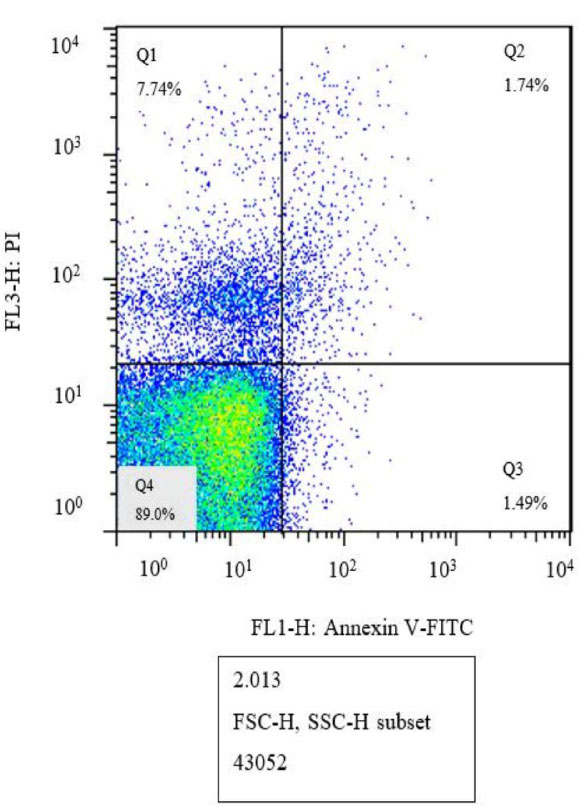
Flow cytometry analysis of linezolid at a concentration of 5 μg/mL (Q1: Necrosis, Q2: Late apoptosis, Q3: Early apoptosis, Q4: Live cells).
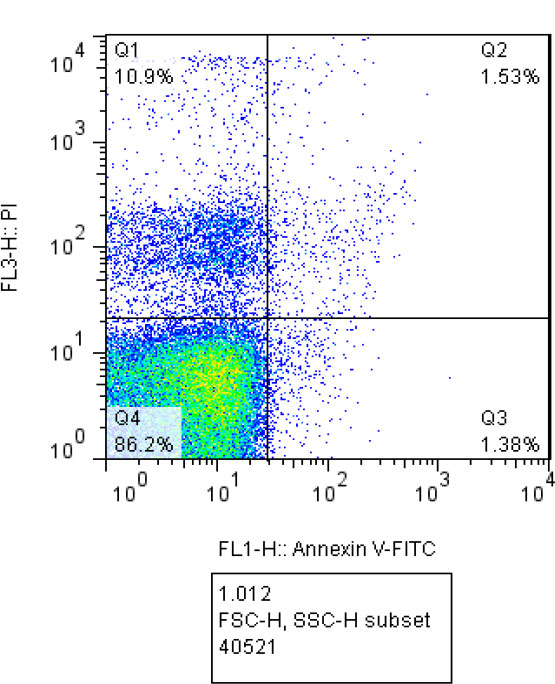
Flow cytometry analysis of linezolid at a concentration of 10 μg/mL (Q1: Necrosis, Q2: Late apoptosis, Q3: Early apoptosis, Q4: Live cells).
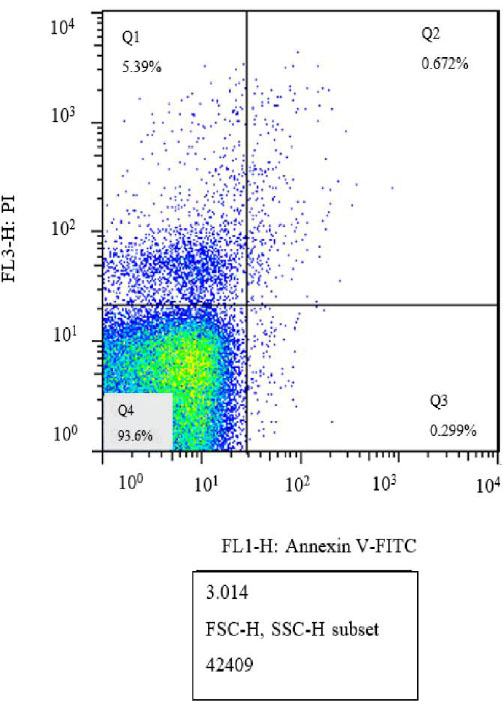
Flow cytometry analysis of the control group without treatment (Q1: Necrosis, Q2: Late apoptosis, Q3: Early apoptosis, Q4: Live cells).
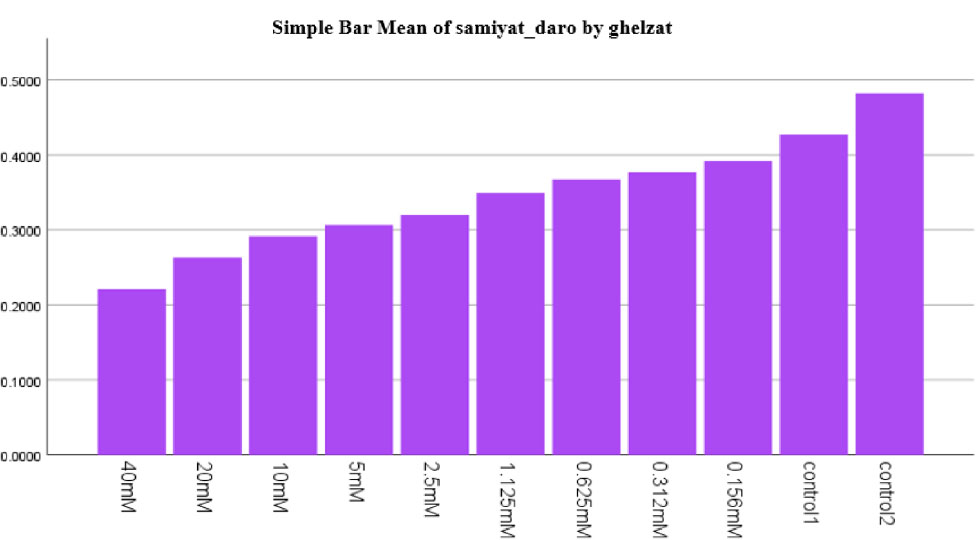
MTT assay of uninfected HeLa cells in the presence of different concentrations of linezolid after 24 hours of incubation in comparison to control groups (control 1 is a positive control and control 2 is a negative control). There are statistical differences with the treated groups in concentration 40 to 5 µg/mL with the negative control group, p<0.05.
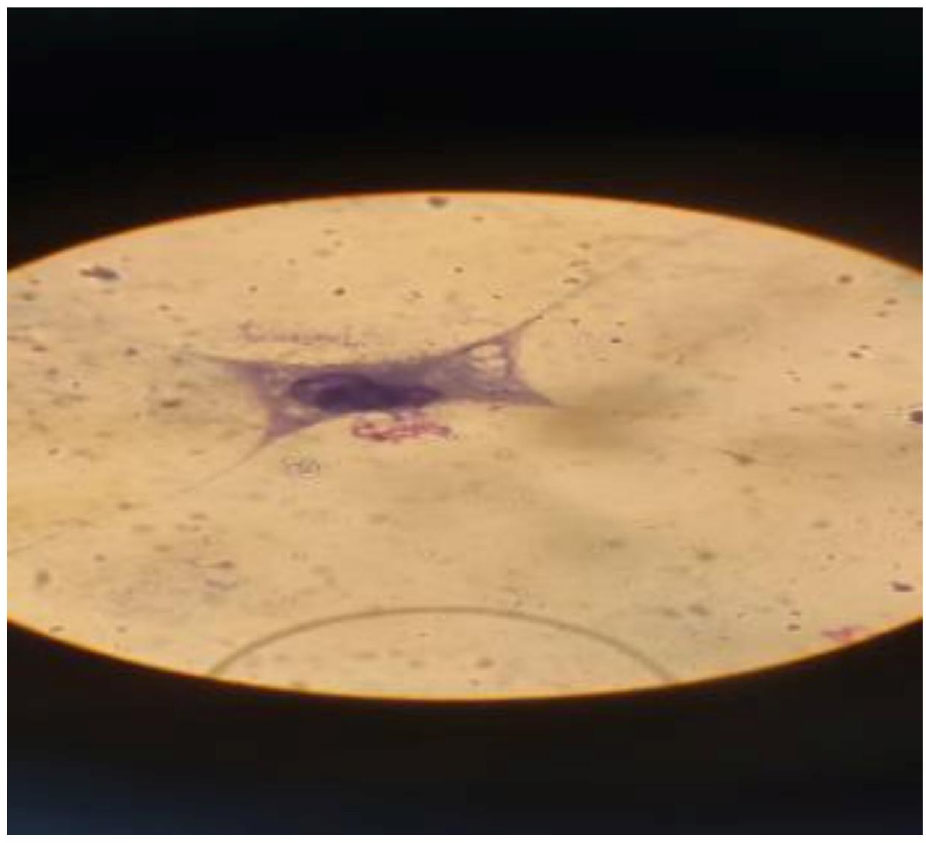
Infected and intact cells in HeLa cells in cell culture by microscope 100X.
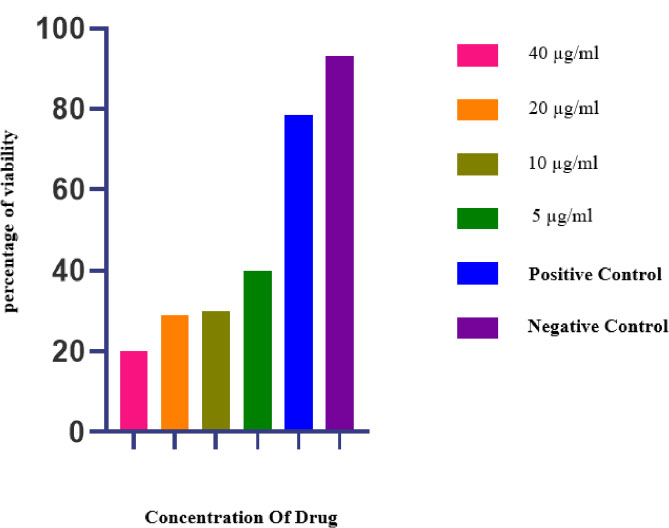
Percentage of parasite-infected cells 24 hours after drug exposure compared to control groups. There are statistical differences between the treated groups with Linezolid, with the negative control group, p<0.05.
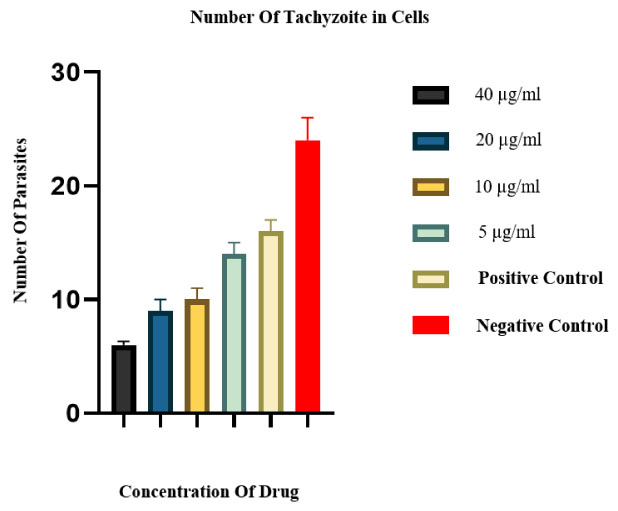
Mean and standard deviation of tachyzoite counts in treated and control cells after 24h. There are statistical differences for all treated groups in comparison to the negative control group p<0.05.
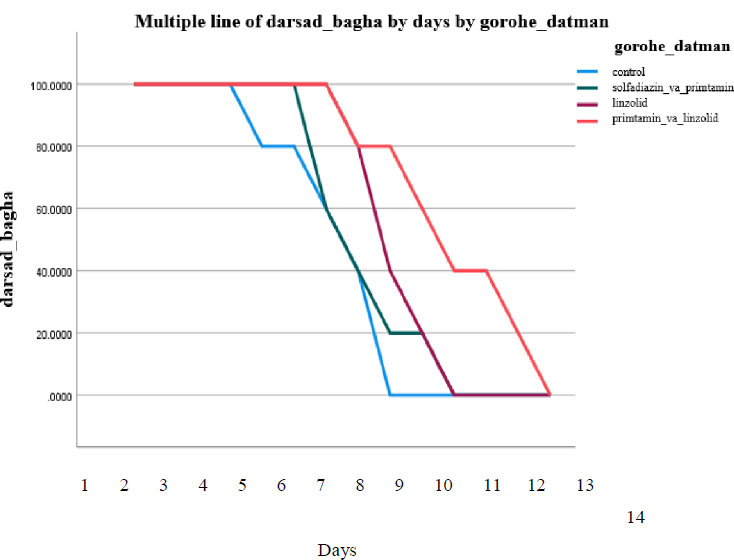
Survival percentage of different treated groups and controls in mice infected with Toxoplasma gondii over 14 days. According to the Kaplan–Meier test, there are statistical differences between the treated and the negative control group, p<0.05.
4. DISCUSSION
The present study aimed to evaluate the potential of linezolid as a therapeutic agent against Toxoplasma gondii using both in vitro and in vivo models. The obtained results revealed that linezolid effectively inhibited tachyzoite growth in a dose-dependent manner, as evidenced by a significant decrease in parasite counts at various time points. There are a limited number of studies evaluating the effect of linezolid on protozoa [14]. Linezolid is a synthetic antibiotic that inhibits microbial protein synthesis by binding to rRNA on both the 30S and 50S ribosomal subunits. The medication prevents the formation of the initiation complex, which can reduce the length of the extended peptide chains and slow down the rate of the translation reaction [15]. However, the initiation process at the site of inhibition takes place prior to that of other protein synthesis inhibitors that prevent the elongation procedure [16]. Due to the unique site of inhibition, cross-resistance to other protein synthesis inhibitors has not yet been demonstrated. Linezolid may also inhibit the expression of virulence elements that lead to a reduction in toxins produced by pathogenic microorganisms [17]. The activity of the compound is increased by the morpholino group in the first ring (from the left) and the fluoride atom in the second ring.
Barthel et al. evaluated the efficacy of linezolid on Plasmodium falciparum. Protein synthesis inhibitors such as doxycycline and azithromycin were used as controls, and the antimalarial effects of these antibiotics were attributed to their activity on prokaryotic organelles, such as mitochondria and apicoplasts. However, linezolid was found to be less effective than the other protozoa [18]. Lackner et al. reported that in an immunocompromised patient with acute granulomatous Acanthamoeba encephalitis, combination therapy with linezolid, meropenem, moxifloxacin, and fluconazole was effective in improving survival [19]. In the present study, flow cytometry analysis also demonstrated the drug's mechanism, revealing that linezolid induces apoptosis in tachyzoites, with distinct populations identified as undergoing primary and secondary apoptosis, as well as necrosis. Ahmadpour et al. indicated the parasite’s ability to interfere with host cell death pathways, such as apoptosis, necrosis, and autophagy, through the modulation of gene expression and the production of microbicidal molecules such as reactive oxygen species (ROS) and nitric oxide (NO) [20]. They reported that the variability in host cell responses to T. gondii is characterized by differences in the expression of immune-related proteins across cell lineages [21]. Oxidative stress arises when the cell’s redox balance is disrupted. It has been proposed that the widespread suppression of mitochondrial activity under such conditions may promote apoptosis due to the resulting sharp reduction in cellular energy production. The results obtained from this study confirmed the results of the present study. Furthermore, the MTT assay in the present study showed that linezolid did not affect the viability of uninfected HeLa cells at the concentrations tested, indicating its selective toxicity towards T. gondii. This selectivity is crucial for therapeutic applications, as it minimizes potential side effects on host cells. Abbasali et al. evaluated the efficacy of the antimicrobial peptide CM11 against Toxoplasma gondii infection in vitro and in vivo, highlighting its potential as an alternative treatment for toxoplasmosis. In their study, the MTT assay showed that CM11 had a cytotoxic effect on VERO cells with a CC50 value of 17.4 μg/mL, while sulfadiazine (SDZ) had a CC50 value of 62.3 μg/mL. The IC50 values for CM11 and SDZ in infected cells were 1.9 μg/mL and 1.4 μg/mL, respectively, indicating that both compounds effectively reduced tachyzoite viability [22]. Their study indicated that CM11 induced apoptosis in tachyzoites and uninfected VERO cells, with higher rates observed at increasing concentrations. In vivo, treatment with CM11 and its combination with SDZ resulted in increased survival time in infected mice [22]. The results of the current study showed that CM11 was effective against T. gondii and could serve as a promising therapeutic agent for toxoplasmosis, potentially minimizing side effects on host cells. These results were consistent with those from previous studies.
In the in vivo part of the present study, the survival of BALB/c mice treated with linezolid, although limited, was significant compared to the control group, which died on day 9. The results revealed that the combination treatment of pyrimethamine and linezolid increased survival beyond that of either drug alone. The survival rate observed in the present study demonstrated the need to investigate new therapeutic strategies against T. gondii, especially for immunocompromised patients. The higher survival of mice treated with the combination of pyrimethamine and linezolid compared to other groups revealed that linezolid could serve as a valuable adjunct in the treatment of toxoplasmosis. Patients with weakened immune systems, such as those living with HIV/AIDS, undergoing chemotherapy or immunosuppressive therapy for autoimmune diseases, are at increased risk for opportunistic infections, including toxoplasmosis [23]. Traditional treatments, such as pyrimethamine and sulfadiazine, have shown efficacy but are often limited by side effects and resistance problems [24]. The survival observed when combining pyrimethamine with linezolid in this study suggested that adjuvant therapies can play an important role in improving outcomes for these vulnerable groups. This study investigated the potential of linezolid, a well-known antibiotic, as an alternative therapeutic agent against T. gondii, which had not been fully investigated before. This contributes to the search for new therapeutic options for a major global health concern. In this study, a combination of both in vitro and in vivo methods provided a robust framework for evaluating the efficacy of linezolid. In addition, the use of different concentrations and multiple evaluation methods (growth inhibition assays, flow cytometry, and MTT assay) provided depth to the findings. The in vivo evaluation lasted only 14 days, which may not fully reflect the long-term effects and potential development of T. gondii resistance to linezolid. The study also used only the BALB/c mouse model. Besides, the results may not be generalizable to other strains or species, and additional models may be needed to evaluate the broader utility of linezolid. Our findings demonstrated that linezolid significantly inhibited tachyzoite viability, reduced intracellular parasite burden, and induced apoptosis in tachyzoite, with moderate extension of survival in infected BALB/c mice. These results collectively suggest that linezolid possesses measurable antitoxoplasma activity, although not superior to standard therapies, and may hold potential as an adjunct agent. We can also suggest comparing the findings with similar repurposing attempts of antibiotics such as azithromycin, clindamycin, and doxycycline against T. gondii or other apicomplexans. We also mention the limitations of this study, such as a small sample size, the absence of a chronic infection model, and the need for higher-dose pharmacokinetic analysis and molecular docking for this drug against T. gondii.
CONCLUSION
This study highlights the potential of linezolid as a therapeutic candidate against T. gondii. Our in vitro findings demonstrated significant inhibition of tachyzoite growth and induction of apoptosis, with minimal toxicity to uninfected HeLa cells. In vivo, linezolid treatment improved survival in BALB/c mice, with the longest survival observed when combined with pyrimethamine. These results indicate that linezolid may serve as a promising adjunct therapy for toxoplasmosis.
However, confirmation of its therapeutic value will require studies with longer treatment durations, larger sample sizes, and additional animal models, including chronic infection systems. Future research should also investigate the molecular mechanisms underlying its antiparasitic effects and evaluate potential synergistic interactions with existing standard treatments.
AUTHORS’ CONTRIBUTIONS
It is hereby acknowledged that all authors have accepted responsibility for the manuscript's content and consented to its submission. They have meticulously reviewed all results and unanimously approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| T. gondi | = Toxoplasma gondi |
| PBS | = Phosphate-Buffered Saline |
| ROS | = Reactive Oxygen Species |
| DZ | = Sulfadiazine |
| SD | = Standard Deviation |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study protocol received approval from the Ethics Committee of Tarbiat Modares University, with the study being assigned the code of ethics IR.MODARES.AEC. 1402.065.
HUMAN AND ANIMAL RIGHTS
The study followed the US National Research Council's Guide for the Care and Use of Laboratory Animals.
This study adheres to internationally accepted standards for animal research, following the 3Rs principle. The ARRIVE guidelines were employed for reporting experiments involving live animals, promoting ethical research practices
AVAILABILITY OF DATA AND MATERIALS
The data and supportive information are available within the article.
ACKNOWLEDGEMENTS
This work was supported by Tarbiat Modares University. This organization is hereby appreciated.


